Multiple Sclerosis > Difficult Diagnoses
A Practical Guide to Diagnosing & Undiagnosing Multiple Sclerosis
A 3-step approach for the busy clinician.
Copy Link
Misdiagnosis of multiple sclerosis (MS) is common and occurs chiefly for 1 of the following 5 reasons:
1. There is no pathognomonic clinical feature or diagnostic test for MS;
2. The differential diagnosis includes a long list of conditions that can be credibly mistaken for MS;1,2
3. The clinical presentations of MS are variable;
4. MS is not common enough for extensive experience with it to accumulate outside specialized centers; and
5. There is pressure to diagnose MS quickly in order to initiate disease-modifying therapy (DMT) early to forestall relapses and long-term disability.3
Despite the accepted McDonald Criteria for MS diagnosis addressing the possibility of misdiagnosis and suggesting strategies to mitigate that risk,4 the rate of MS misdiagnosis remains high and may even be rising. A recent study suggested almost 20% of people referred to MS clinics with a diagnosis of MS did not actually have the disease.5 This would translate to misdiagnosis and possibly inappropriate treatment of 180,000 people considering the current prevalence of MS in the US of approximately 900,000.6
A challenge with implementing the diagnostic criteria is that they should be applied to patients who have experienced a “monophasic clinical episode. . . similar to a typical multiple sclerosis relapse.4” This presupposes expertise in recognizing typical MS relapses, which is generally the hardest part of the diagnostic process. In a recent survey, more than 30% of neurology residents and 15% of MS specialists misidentified syndromes of complete myelopathy and complete gaze palsy as typical of MS relapse,7 even though both of these syndromes were specifically described as inconsistent with MS in the McDonald Criteria.4 Use of the criteria also requires knowledge of size, location, and morphology of demyelinating lesions on MRI. In practice, misclassification of nonspecific vs demyelinating lesions on brain MRI is a leading contributor to MS misdiagnosis.8,9 It is likely that incorporation of the more specific radiographic signs of demyelination into the diagnostic criteria, such as the central vein sign10 or paramagnetic rims,11 will greatly decrease the risk of MRI-supported MS misdiagnosis. Recognition of these signs, however, requires optimization of MRI sequences not yet available outside of a few specialized centers.
A Practical 3-Step Approach
A practical and easy-to-use approach to diagnose (or undiagnose) MS is needed. A proposed tool, shown in the Figure, requires the practitioner to sequentially address the following 3 key questions:
1. Is the clinical presentation suggestive of MS relapse?
2. Are MRI findings consistent with demyelination?
3. Do findings of cerebrospinal fluid (CSF) analysis support MS diagnosis?
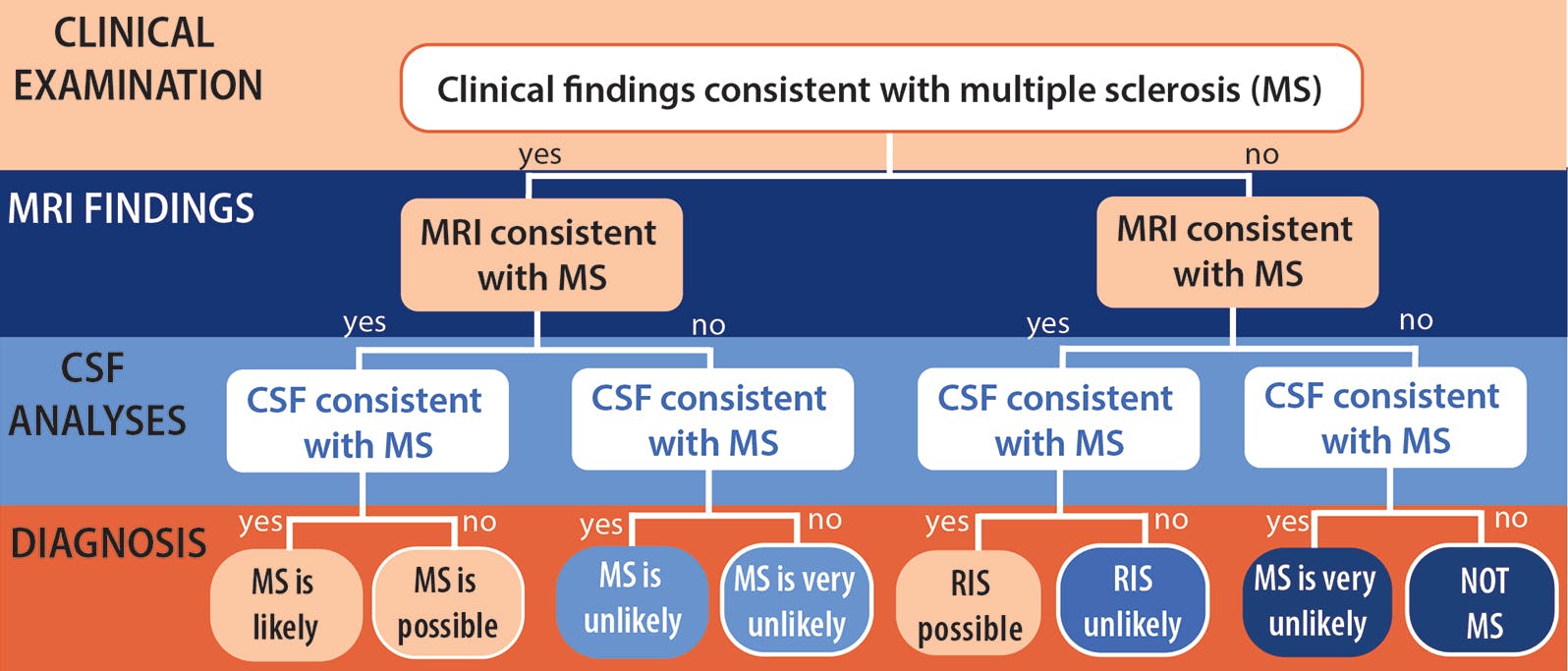
Figure. In the 3-step approach to MS diagnosis, clinicians start with the clinical history and examination and proceed to review of MRI and then CSF findings. The probability of a diagnosis of MS depends on which path through the steps is followed for an individual patient. Abbreviations: CSF, cerebrospinal fluid; RIS, radiologically isolated syndrome.
Step 1. Clinical Assessment for MS Relapses
Whether the clinical presentation is suggestive of an MS relapse depends on 2 main factors: timing and localization of symptoms. MS relapses are typically caused by expanding foci of inflammation that disrupt signal transmission along large, densely myelinated white matter tracts.a
Thus, symptoms typically increase in intensity over days to weeks, followed by slow and often incomplete resolution. Even at their peak, symptoms rarely cause complete loss of function. Thus, blindness, complete gaze palsy, paraplegia are red flags for MS diagnosis. Symptoms can usually be localized to specific white matter tracts (Table).
Timing of symptoms should be ascertained by careful history taking. Localization should be determined by eliciting relevant signs on neurologic examination, with the caveat that the examination outside of the acute relapse period is usually normal in early MS. A history of 1 or more relapse-like episodes suggests MS should be high on the differential, whereas the absence of relapses makes MS unlikely. An important exception to that rule is when a person presents with progressive myelopathy plus syndrome (ie, progressive ambulation and autonomic dysfunction), often accompanied by cognitive, affective, visual, and cerebellar signs that tend to be less prominent than myelopathy. This presentation is typical of the primary progressive subtype of MS, which affects about 10% of MS patients. Progressive MS can be diagnosed only in retrospect, after at least 12 months of neurologic deterioration.
Importantly, symptoms that neither localize to specific white matter tracts nor follow the typical temporal progression of MS, such as headache, cognitive problems (often described as brain fog), fatigue, intermittent tingling in different parts of the body, dizziness, myalgias, or arthralgias, do not provide clinical evidence suggestive of MS despite being very common in MS.
Step 2. MRI Assessment for Demyelination
Whether lesions seen on MRI are suggestive of MS is of utmost importance. Misidentification of lesions as demyelinating is a key driver in misdiagnosis. My prior publication, The MS Lesion Checklist, outlines a practical approach to MS lesion identification,14 and Filippi et al provide a more formal, in-depth discussion.15 The main utility of The MS Lesion Checklist is that it allows rapid screening of brain MRI for lesions typical of demyelination. If the MRI does not show the telltale signs of demyelination, the diagnosis of MS is highly unlikely. As a rule, people whose clinical history is not suggestive of MS will not have MRI findings that are suggestive of MS. However, a small proportion of people with nonspecific neurologic symptoms, or no symptoms at all, do have characteristic demyelinating lesions on brain MRI and may develop clinical MS over time—a scenario known as radiologically isolated syndrome.16
Step 3. CSF Analysis for Neuroinflammation
CSF analysis is not required for MS diagnosis, but it can be extremely helpful when clinical and MRI assessments are equivocal. Multiple unmatched oligoclonal bands (OCBs) in the CSF (ie, bands found in CSF but not serum) are found in more than 90% of people with MS. Absence of OCBs makes nonMS diagnoses 15 times more likely than when OCBs are present.2
There are 2 critical caveats for interpreting CSF findings. First, matched OCBs (ie, identical bands found in both CSF and serum samples) indicate systemic inflammation and are evidence against a diagnosis of MS. Second, unmatched OCBs are not specific for MS because they are found in dozens of other disorders, particularly chronic CNS infections and neuroinflammatory disorders.17 The number of OCBs is generally higher in MS than in other diseases: more than 9 OCBs were found in 46% of people with MS, but only 14% of other disorders.18 The cut-off of 3 OCBs has the advantage of increased specificity without sacrificing too much sensitivity, but some laboratories report a positive OCB result when only 1 to 2 OCBs are present, and sometimes even when matched OCBs are present. It is essential to read the fine print when interpreting OCB results. Elevated IgG index in CSF is another sign of intrathecal inflammation but it is not as sensitive for MS as OCBs. CSF analyses should also be examined for findings that are inconsistent with MS, such as lymphocyte count over 50 cells/mm3; the presence of neutrophils, eosinophils or atypical cells; and protein concentration over 100 mg/dL. Any of these features cast strong doubt on MS diagnosis and can be decisive in ruling out MS when clinical and MRI features are not otherwise convincing.
Conclusion
The question “Could this be MS?” arises frequently in neurologic practice. To answer this question with confidence, the clinicians need to ask themselves whether clinical history is consistent with MS relapse or progression; whether MRI discloses lesions suggestive of demyelination; and whether CSF analyses show multiple unmatched OCBs. When clinical symptoms do not localize to a commonly affected myelinated tracts and do not follow a temporal pattern of evolution consistent with an inflammatory process, the likelihood of MS is low. The absence of MS-typical lesions in such patients makes the diagnosis of MS untenable. In equivocal cases, CSF studies can be very helpful as the absence of OCBs is a major predictor of non-MS diagnosis. Discussion of alternatives to MS diagnosis lie outside the purview of this article, but comprehensive reviews of this topic are freely available.1,19
1. Charil A, Yousry TA, Rovaris M, et al. MRI and the diagnosis of multiple sclerosis: expanding the concept of “no better explanation”. Lancet Neurol. 2006;5(10):841-852.
2. Calabrese M, Gasperini C, Tortorella C, et al. “Better explanations” in multiple sclerosis diagnostic workup: a 3-year longitudinal study. Neurology. 2019;92(22):e2527-e2537. doi: 10.1212/WNL.0000000000007573
3. Solomon AJ, Corboy JR. The tension between early diagnosis and misdiagnosis of multiple sclerosis. Nat Rev Neurol. 2017;13(9):567-572.
4. Thompson AJ, Banwell BL, Barkhof F, et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 2018;17(2):162-173.
5. Kaisey M, Solomon AJ, Luu M, Giesser BS, Sicotte NL. Incidence of multiple sclerosis misdiagnosis in referrals to two academic centers. Mult Scler Relat Disord. 2019;30:51-56.
6. Wallin MT, Culpepper WJ, Campbell JD, et al. The prevalence of MS in the United States: a population-based estimate using health claims data. Neurology. 2019;92(10):e1029-e1040. doi: 10.1212/WNL.0000000000007035.
7. Solomon AJ, Pettigrew R, Naismith RT, Chahin S, Krieger S, Weinshenker B. Challenges in multiple sclerosis diagnosis: misunderstanding and misapplication of the McDonald criteria. Mult Scler. 2020:1352458520910496. doi:10.1177/1352458520910496
8. Carmosino MJ, Brousseau KM, Arciniegas DB, Corboy JR. Initial evaluations for multiple sclerosis in a university multiple sclerosis center: outcomes and role of magnetic resonance imaging in referral. Arch Neurol. 2005;62(4):585-590.
9. Solomon AJ, Bourdette DN, Cross AH, et al. The contemporary spectrum of multiple sclerosis misdiagnosis: a multicenter study. Neurology. 2016;87(13):1393-1399.
10. Gaitán MI, Yañez P, Paday Formenti ME, et al. SWAN-Venule: an optimized MRI technique to detect the central vein sign in MS plaques. AJNR Am J Neuroradiol. 2020;41(3):456-460.
11. Maggi P, Sati P, Nair G, et al. Paramagnetic rim lesions are m sclerosis: an international multicenter 3T MRI study. Ann Neurol. 2020;88(5):1034-1042.
12. Fisher E, Lee JC, Nakamura K, Rudick RA. Gray matter atrophy in multiple sclerosis: a longitudinal study. Ann Neurol. 2008 Sep;64(3):255-265.
13. Schumacher GA, Beebe G, Kibler RF, et al. Problems of experimental trials of therapy in multiple sclerosis: report by the Panel on the Evaluation of Experimental Trials of Therapy in Multiple Sclerosis. Ann NY Acad Sci 1965;122:552-568.
14. Kister I. The Multiple Sclerosis Lesion Checklist: a clinician’s guide to evaluating brain MRI in a patient with improbable multiple sclerosis. Pract Neurol (US). 2018;19(6):68-72.
15. Filippi M, Preziosa P, Banwell BL, et al. Assessment of lesions on magnetic resonance imaging in multiple sclerosis: practical guidelines. Brain. 2019;142(7):1858-1875.
16. Lebrun-Frenay C, Kantarci O, Siva A, et al. Radiologically isolated syndrome: 10-year risk estimate of a clinical event. Ann Neurol. 2020;88(2):407-417.
17. Petzold A. Intrathecal oligoclonal IgG synthesis in multiple sclerosis. J Neuroimmunol.2013;262(1-2):1-10.
18. Bourahoui A, De Seze J, Guttierez R, et al. CSF isoelectrofocusing in a large cohort of MS and other neurological diseases. Eur J Neurol. 2004;11(8):525-529.
19. Miller DH, Weinshenker BG, Filippi M, et al. Differential diagnosis of suspected multiple sclerosis: a consensus approach. Mult Scler. 2008;14(9):1157-1174.
Disclosure
IK has disclosures at www.theneurologyhub.com





